Heart failure, a condition that arises from a variety of structural and functional cardiac diseases, remains a significant health challenge with a poor prognosis. A recent study published in Scientific Reports highlights the deterioration of heart function associated with nicotine exposure, pinpointing molecular mechanisms that could potentially inform new therapeutic strategies. Nicotine, a prominent alkaloid found in tobacco, has been implicated in the pathological processes that lead to myocardial remodeling, a key precursor to heart failure.
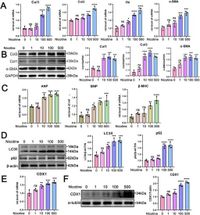
The research delves into the precise biological activities of CDX1, a transcription factor recently found to play a critical role in cardiac fibroblast activation and cardiomyocyte hypertrophy when stimulated by nicotine. According to the authors, CDX1 expression is increased in response to nicotine, suggesting that it may attempt to mitigate damage from this toxic substance. However, they also found that when CDX1 levels drop, there is a subsequent aggravation of fibroblast activation and cardiac hypertrophy due to impaired autophagic flux.
In their findings, the researchers observed that elevated CDX1 expression counteracts the blockade of autophagic flux, a vital cellular process that helps regulate the degradation and recycling of cellular components. This blockade, when manipulated to favor the activation of the mechanistic target of rapamycin (mTOR) pathway, exacerbates harm to cardiac cells, ultimately promoting heart failure.
Specifically, the study shows that nicotine induced a concentration-dependent upregulation of fibrosis markers like Col1, Col3, and alpha-SMA in cardiac fibroblasts (CFs) and hypertrophy markers such as ANP, BNP, and β-MHC in neonatal rat ventricular myocytes (NRVMs). After nicotine treatment, the activation of CFs and hypertrophy of NRVMs was evident, confirming that the harmful effects of nicotine extend beyond the immediate damage to heart muscle cells and influence surrounding fibroblasts.
To understand the underlying molecular relations, the researchers conducted further investigations on the autophagy process, which is critical for cellular homeostasis and function. They found that silencing CDX1 significantly increased levels of autophagic substrates such as LC3II and p62 under nicotine stimulation, indicating that CDX1 is essential for maintaining autophagic flux.
In contrast, overexpressing CDX1 helped alleviate this flux blockade while enhancing LAPTM4B, a gene implicated in the regulation of autophagy. "Our study demonstrates that CDX1 promotes the transcription of LAPTM4B," wrote the authors, thereby presenting a novel mechanism by which CDX1 may protect cardiac cells from nicotine-induced damage.
Further experiments revealed that downregulation of CDX1 not only increased CF activation but also heightened cardiomyocyte hypertrophy when stimulated by nicotine. The findings showed that LAPTM4B, expressed at elevated levels in cardiac tissue post-nicotine exposure, plays a pivotal role in modulating autophagic flux, suggesting a direct transcriptional link with CDX1.
Interestingly, the authors highlighted the balance of mTOR pathway activity. mTORC1 is known to inhibit autophagy, and the researchers found that CDX1 could reverse nicotine's activation of mTOR, particularly its downstream effectors such as p70S6K and 4EBP1. This suggests that by moderating the mTOR pathway, CDX1 and LAPTM4B might mitigate some of the harmful effects of nicotine on heart function.
The researchers used a combination of techniques including siRNA knockdown and adenoviral vector transfection to detail how modulation of CDX1 impacts autophagic processes. For instance, they documented that downregulation of LAPTM4B exacerbated nicotine-induced damage, mirroring the detrimental effects seen with CDX1 suppression.
The implications of this research are far-reaching, as nicotine is well-known to be a significant risk factor for cardiovascular disease. With heart failure projected to affect millions in the coming decades, understanding the mechanistic pathways by which nicotine induces cardiac impairments is crucial for developing preventative strategies. By illuminating the protective roles of CDX1 and LAPTM4B within these pathways, the study opens new avenues for therapeutic intervention and could inform the development of targeted treatments aimed at combating the cardiovascular risks associated with smoking.
In summary, this research provides compelling insights into how the transcription factor CDX1 and its regulatory role on LAPTM4B help to restore autophagic flux in the presence of nicotine, offering a potential cardioprotective function and valuable perspective on combating heart failure driven by tobacco use.