The interaction between neutrophil elastase (NE) and Toll-like receptor 4 (TLR4) has garnered significant scientific interest, particularly regarding its implications in cardiovascular diseases. A recent study has validated this interaction using advanced computational tools and experimental models, offering new insights that could inform therapeutic strategies in managing cardiovascular conditions.
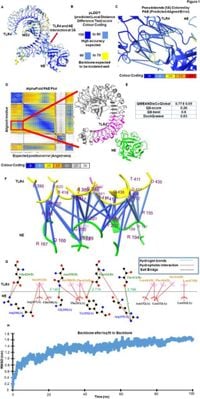
Researchers from Jamia Hamdard, New Delhi, employed state-of-the-art technology, including the AlphaFold2 program, which utilizes deep learning to predict protein structures, to explore how rat NE binds with rat TLR4. Their findings strongly suggest a potential interaction between these proteins, which could play a significant role in the progression of cardiovascular diseases.
Neutrophils are vital components of the innate immune system, acting quickly to migrate to areas of inflammation or infection to aid in tissue repair. During this immune response, NE, a serine protease, plays a crucial role not only in combating pathogens but also in contributing to various cardiovascular ailments. Previous research has linked NE with conditions like cardiac hypertrophy and atherosclerosis, while TLR4 has been associated with immune responses and inflammation in similar contexts.
In their study, the team observed elevated levels of NE and TLR4 in cardiac tissues after inducing hypertrophy in rat models using isoproterenol (ISO). This induction mimics heart conditions that lead to complications such as heart failure. Employing co-immunoprecipitation techniques alongside the computational predictions, the researchers confirmed the binding of NE and TLR4, highlighting a molecular interplay that had not been previously established.
Computational analyses further validated these findings. The AlphaFold2 predictions indicated that rat NE binds to the central region of rat TLR4's extracellular domain, establishing a distinct interaction most likely pivotal to cardiovascular disease pathology. Notably, prior to this study, while evidence suggested an association between NE and TLR4, a direct interaction had not been demonstrated. Thus, the research adds a new layer to the understanding of cardiovascular responses to neutrophil activity.
Furthermore, the study showcases the efficacy of AlphaFold2 as a robust tool in structural biology, capable of accurately predicting protein interactions. This could have far-reaching implications for future cardiovascular therapies aimed at inhibiting such binding interactions. As the study highlights, targeting this NE-TLR4 interaction may open up promising avenues for developing interventions that can mitigate inflammation's role in cardiovascular health.
Importantly, heightened levels of NE have been previously associated with adverse cardiac events like heart failure and myocardial infarctions. The correlation between NE’s activity and cardiovascular health underscores the necessity for ongoing research into these pathways, particularly as investigators explore therapeutic options that could improve outcomes for patients suffering from heart diseases.
In conclusion, this innovative research provides compelling evidence for the binding interaction between NE and TLR4, both through computational predictions and robust experimental validation. The implications of this work suggest that not only does it advance our understanding of cardiac hypertrophy but also points to the potential for new therapeutic strategies targeting this molecular interaction.